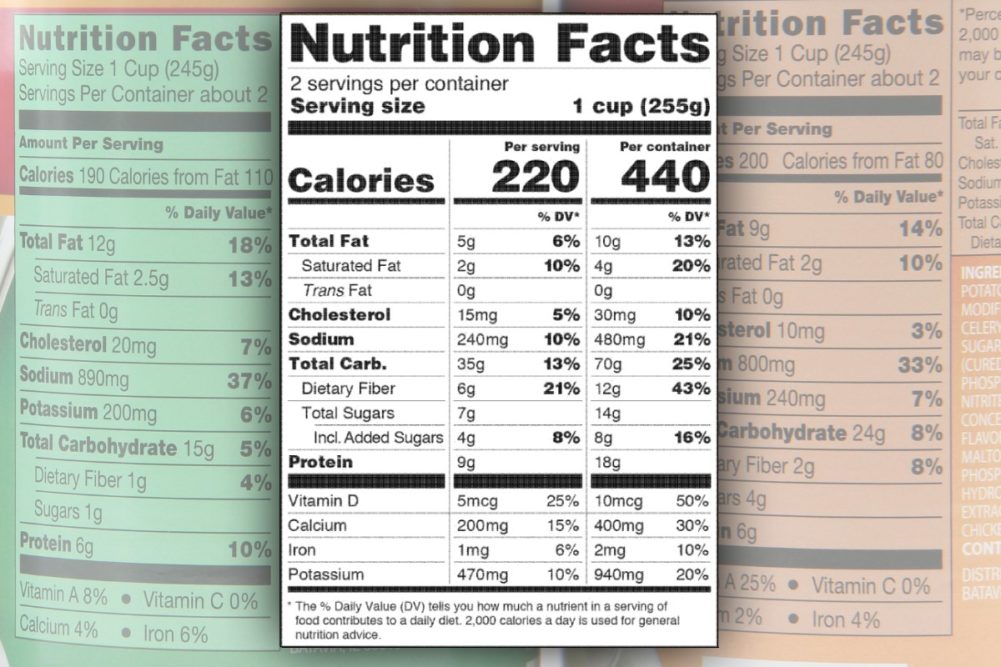
SILVER SPRING, MD. — The US Food and Drug Administration (FDA) has issued additional information about requirements for dual column labeling and serving sizes on the Nutrition Facts Label. The information is being provided in response to questions posed by stakeholders in the food industry.
Since the introduction of the new Nutrition Facts Label, some products have featured dual column labeling if they can reasonably be consumed in one meal or snack. Products that fall under that category have separate labels listing the nutrition facts for a single serving and nutrition facts for the contents of the entire package.
The FDA hopes its latest information will help clear up confusion.
“In the guidance we issued today, we are helping answer some of the questions most commonly asked by industry about dual column label and serving size issues, including when dual column labeling is required, the definition of a single-serving container, how to determine the number of servings in a food product, and how these labels should be formatted for some products that have limited space for nutrition labeling,” said Claudine Kavanaugh, Ph.D., RD, director of the Office of Nutrition and Food Labeling in the FDA’s Center for Food Safety and Applied Nutrition. “The new Nutrition Facts Label has updated serving sizes for many foods. We know that Americans are eating differently, and the amount of calories and nutrients on the label is required to reflect what people actually eat and drink — not a recommendation of what to eat or drink. The new label, including this dual column layout, will drive consumers' attention to the calories and percent daily value of nutrients that they are actually consuming.”
Manufacturers with $10 million or more in annual food sales are required to comply with the updated Nutrition Facts Label regulations as of Jan. 1, 2020, while manufacturers with less than $10 million in annual food sales will have an additional year to comply. During the first six months following the Jan. 1, 2020, compliance date, the FDA said it plans to work cooperatively with manufacturers to meet the new Nutrition Facts Label requirements and will not focus on enforcement actions regarding these requirements during that time.